Microbial Retention Testing Under Liquid and Aerosol Conditions: 3 Techniques
September, 2023
Microbial retention is the single most important performance attribute of sterilizing-grade air and gas filters. In the food and beverage industry, the sterility of air or gas is crucial to the success of filtration processes to ensure the end product is contaminant-free.
Understanding how sterilizing-grade cartridge air filters are validated for microbial removal from air or gas assists the end user in effective filter selection. According to guidance from FDA, the validation work carried out by filter manufacturers demonstrates the microbial retention and removal efficiency of hydrophobic sterilizing-grade cartridge air filters when challenged with a defined minimum concentration of model microorganisms, namely, Brevundimonas diminuta (B. diminuta) (ATCC 19146) at 107 cfu/cm2 of effective filtration area, either from a liquid suspension (“liquid bacterial challenge”) or from a gas (“aerosol bacterial challenge”), or both.
In addition to these basic bacterial challenge tests, which sufficiently demonstrate the sterilizing ability of cartridge air filters, filter manufacturers may do more validation work demonstrating filter retention of bacterial spores, long-term aerosol bacterial challenges, and aerosol bacteriophage removal, to illustrate the filters’ fit for purpose in various applications.
Our team at Pall leverages three ways to validate microbial retention performance of filters. Below are three methods used by filter manufacturers to validate microbial retention performance.
1) Liquid and Gas Removal Performance in Air Filtration
Filters perform differently based on whether they are removing contaminants from a liquid or a gas. Filters have both a liquid and a gas removal rating. Due to the mechanisms at play in liquid or gas filtration, it is more “difficult” to remove contaminants from liquids than from gases, i.e., it requires a “tighter” filter to remove contaminants from a liquid than from a gas. Further, a liquid-rated 0.2 µm filter, which removes the described contaminants from a liquid, is much tighter than a gas-rated 0.2 µm filter, which removes these contaminants from a gas.
The bacterial challenge test is conducted under normal (gas) and worst-case (liquid) lab conditions simulating production conditions.
2) Liquid Bacterial Challenge and Validation Testing
Liquid bacterial challenge demonstrates the filter’s microbial retention and removal efficiency performance when challenged with a liquid suspension inoculated with the challenge microorganism.
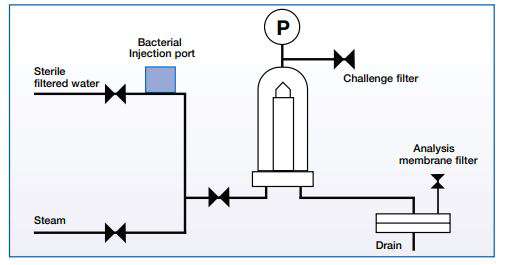
Figure 1 illustrates a typical liquid bacterial challenge test set-up based on guidance from ASTM F838-83 and updated versions. If even one viable microorganism is found, the filter has failed the validation test. Another filter integrity test is carried out to reconfirm filter integrity after the challenge test is completed.
3) Aerosol Bacterial Challenge Validation Testing
Aerosol bacterial challenge demonstrates the filter’s microbial retention and removal efficiency performance when challenged in a gas with the nebulized challenge microorganism. Figure 2 illustrates a typical aerosol bacterial challenge setup. There are different approaches available to conduct this test; the test design must match the purpose for which the filter is being tested.
Comparison of Liquid Bacterial Challenge and Aerosol Bacterial Challenge Validation
Both microbial validation tests demonstrate the gas sterilizing capability of a filter according to the definition for sterilizing-grade cartridge gas filters, however under different conditions. Both methods provide extremely high sensitivity by evaluating the totality of filter effluent, they can detect even very low numbers of viable bacterial cells which may have penetrated the filter.
Validating sterile filtration of gases that come into contact with products or packaging is a critical component to ensuring the filtration process is fit for purpose so that the end product is contaminant-free. You can learn more about the details of each validation test in our full article.
To download the full bulletin, complete the form below and submit
- Category
- Author
- Sort By